Bringing genomics into the fold--the protein fold
With a five-year, (2020-2025) $1.95 million National Institutes of Health R35 grant, the Brandizzi Lab in the College of Natural Science is expanding its exploration of protein folding within plant cells, paving the way to more effective treatments of human diseases caused by misfolded proteins such as diabetes, Alzheimer’s and cancer.
Using advanced chemical genomics and gene network analyses, the team will characterize the signaling pathways of the endoplasmic reticulum (ER) that, among other things, monitors protein folding inside cells.

“The endoplasmic reticulum is an amazing biosynthetic factory that produces one-third of the cellular proteome, including antibodies, lipids and proteins,” said Federica Brandizzi, MSU Foundation Professor in the Department of Plant Biology, MSU-DOE Plant Research Laboratory (PRL) faculty member, and lead investigator on the grant. “The proteins we eat in cereals and beans come from the ER where it synthesizes the storage protein important in nutrition.”
Every fungus, plant and animal on earth is dependent on the ER’s three-dimensional spider web of protein producing tubules for survival. The constantly shape-shifting properties of the ER chaperone and fold proteins that can only do their job of growing and protecting the cell if they are folded into the right shape.
Like a master of origami, when the ER senses the excess material of a misfolded protein, it triggers a system that attempts to correct it.
“The ER communicates with the nucleus and manages to re-program gene expression to send helpers towards the ER that make sure it starts folding proteins properly,” Brandizzi explained. “If this system fails, another system is activated that leads to cell death rather than sending out malfolded proteins.”
This sophisticated and largely unexplored monitoring system of the ER is known as the Unfolded Protein Response (UPR), and this is what the Brandizzi Lab has been so interested and adept at understanding for the last 14 years.

To study the genetically important pathways of the UPR requires methods not feasible with humans on the one hand or with simple organisms like fungi on the other. So the Brandizzi Lab uses the evolutionarily distinct Arabidopsis thaliana, a richly studied organism with cells that function similarly to human cells.
“In the lab setting at PRL we can do so much to inform plant and human biological studies,” Brandizzi explained. “With Arabidopsis, we can generate crosses and multiple orders of mutants with a disrupted UPR that we then test in the lab using seed material and plants grown on a plate.”
In previous Arabidopsis studies, Brandizzi’s team conducted novel, forward genetic screens to uncover key steps governing the ER during plant growth and stress as well to locate and characterize proteins critical to the ER’s stress response. They established that loss of the UPR can be lethal under such stressful conditions.
Under the new grant, the team of scientists will develop and analyze a mutant population of Arabidopsis seeds produced from a mutant Arabidopsis plant with a UPR that continues folding proteins under stress—a normally lethal condition.
“The plants all die under stressful conditions, but with the mutations that we introduce into the plant’s genetic code, we can restore UPR functioning phenotypes and allow plants to grow and survive stress,” Brandizzi explained. “Essentially, we bring the plants back to life like zombies.”
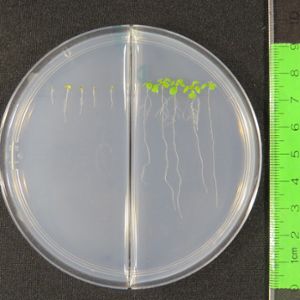
Unlocking the genetic basis for the UPR’s ability to correct folding mistakes as they grow under stress may hold the key for translational research that will boost plant production and correct similar mistakes in human cells.
“This is really exciting and important for humans,” Brandizzi said. “Human cells that have a defect in the UPR might be able to be brought back to life and functional by introducing single mutations that we are mapping in our lab.”
Brandizzi’s research into how plants can help modify human cells also offers potential ways for humans to modify plant cells.
“By understanding how we can propitiate the folding ability of the ER, we can boost the bio-factory activity of the plant cell to make plants generate products such as insulin and antibodies,” explained Brandizzi.
Brandizzi emphasized that such innovative research would not be possible without the community of scientists at the MSU-DOE Plant Research Laboratory, including Evan Angelos, a Department of Biochemistry and Molecular Biology graduate student working on the grant.
“COVID has hit us hard, but we keep moving forward with this exciting research,” Brandizzi said.
Banner image: A confocal microscopy image of the beautifully dynamic endoplasmic reticulum (ER). Every fungus, plant and animal on earth is dependent on the ER’s three-dimensional spider web of protein producing tubules for survival. Credit: Ethan Thibaut