MSU scientists develop novel simulations to help get PFAS out of soil
Michigan State University chemists are discovering new information to help remediate “forever chemicals” by showing for the first time how they interact with soil at the molecular level.

Credit: Harley J. Seeley
The researchers, Narasimhan Loganathan and Angela K. Wilson in the MSU College of Natural Science, published their findings May 11 online in the journal Environmental Science & Technology.
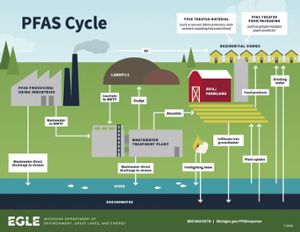
“Forever chemicals” — more formally known as PFAS or perfluoroalkyl and polyfluoroalkyl substances — earned the label because they don’t break down naturally. When PFAS pollute soil and water, they can enter the food system through plants, livestock and drinking water.
A Centers for Disease Control and Prevention report from 2015 estimated that PFAS is in the blood of 97 percent of Americans. Other, more recent studies have put that number closer to 99 percent.
What makes PFAS so ubiquitous is a combination of persistence and utility. More than 9,000 chemicals qualify as PFAS and they’re found in a wide range of applications, including food packaging, nonstick cookware, firefighting foams and many more. While time and nature can degrade certain components of these products — and of the waste generated in producing them — the PFAS lingers, accumulating in the environment.
_250x350.jpg)
Courtesy photo
Removing PFAS from soil and water, then, is important for reducing exposure to these chemicals and the harm they can cause, including thyroid disease and increased risk of some cancers.
“When you start looking at mitigation strategies, you see a lot about removing PFAS from water, but there’s very little about PFAS in soil,” said Loganathan, a senior research associate in MSU’s Department of Chemistry.
“And some of the studies are ‘molecule blind,’” said Wilson, John A. Hannah Distinguished Professor of chemistry and a scientist with the MSU Center for PFAS Research. “That is, they’re not paying attention to the chemistry.”
Wilson and Loganathan decided to help change that by performing the first molecular-level simulations of interactions between PFAS with a soil component, kaolinite.
For the study, the duo focused on some of the most prevalent and problematic PFAS chemicals. They chose kaolinite on the soil side because it is a common soil mineral, especially in Michigan.
PFAS are a concern everywhere, but they present a unique challenge in Michigan. Michigan has an abundance of PFAS, with more than 200 known PFAS-contaminated sites. On top of that, agriculture and the Great Lakes are foundational to the state’s identity. Protecting Michigan’s land and water is a shared goal of many of the state’s communities, legislators and companies.
“Even before this work, we were going to huge meetings and talking about PFAS with people from different municipalities, farms, wastewater treatment plants and more,” Wilson said. “A lot of people are looking for solutions.”
The study was inspired by a Michigan engineering firm that asked Wilson about how PFAS might spread in soil and how best to remediate the chemicals. She didn’t have the answers, but she knew Loganathan could help her start finding some.
She recruited him to join this project, supported by the National Science Foundation. The duo also had access to computational resources provided by the National Energy Research Scientific Computing Center and MSU’s Institute for Cyber-Enabled Research, or iCER.

The results of the simulations did provide some reasons for optimism with regard to remediation. For example, some of the PFAS the researchers studied had longer carbon chains serving as their backbones congregated on the kaolinite.
“Ideally, this is what you’d want. You’d like all PFAS just to sit in a clump so you can grab it and filter it out,” Wilson said. The flipside is that the shorter-chained PFAS were less likely to clump, remaining more mobile in soil.
“The take-home message is that not all PFAS behave similarly,” Wilson said. “And not all soils behave the same with regard to PFAS.”
“The components in the soil play a big role,” Loganathan said. “The soil composition around any contaminated site is going to be critical for how far PFAS make it into the subsurface, where they can then reach groundwater.”
Although the idea of examining the myriad combinations of PFAS and soil components is imposing, the Spartans have shown their computational approach is well-suited to tackling the diversity of problems inherent to PFAS pollution.
“The beauty of computational chemistry is that you can study so many different systems,” said Wilson, whose research team is also examining interactions of PFAS with proteins in the body. Her team is also studying PFAS in different fish species with support from Great Lakes Fisheries Trust and the Strategic Environmental Research and Development Program, which are state and federal organizations, respectively, that fund environmental projects.
The goal, in the soil and biology projects, is to reveal interactions that could help protect more people from PFAS exposure.
“Such molecular level insights are going to be incredibly important for any remediation strategy,” Loganathan said.
Banner image: This image rendering shows several different types of short- and long-chain PFAS molecules the researchers examined to learn how the compounds interact with soil components, such as kaolinite clay. Such comprehensive knowledge of PFAS at the clay mineral interface is critical to developing novel site-specific degradation and mitigation strategies. Credit: Wilson Research Group